
Cancer Therapy Evaluation Program (CTEP)
5 (500) In stock

5 (500) In stock

Common Toxicity Criteria (ctc) - Cancer Therapy Evaluation Program - Ctep Cancer - Fill and Sign Printable Template Online

Cancer Therapy Evaluation Program - an overview

Cracking the Code to CTE

CTIS
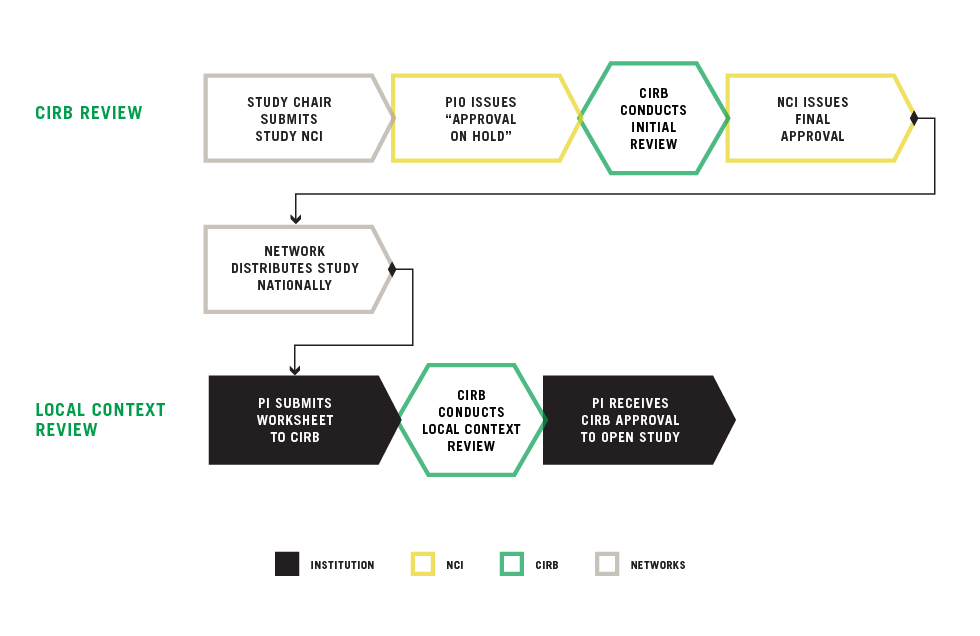
Overview of the Study Review Process

CTEP Interactions with Industry and the FDA - ppt video online download
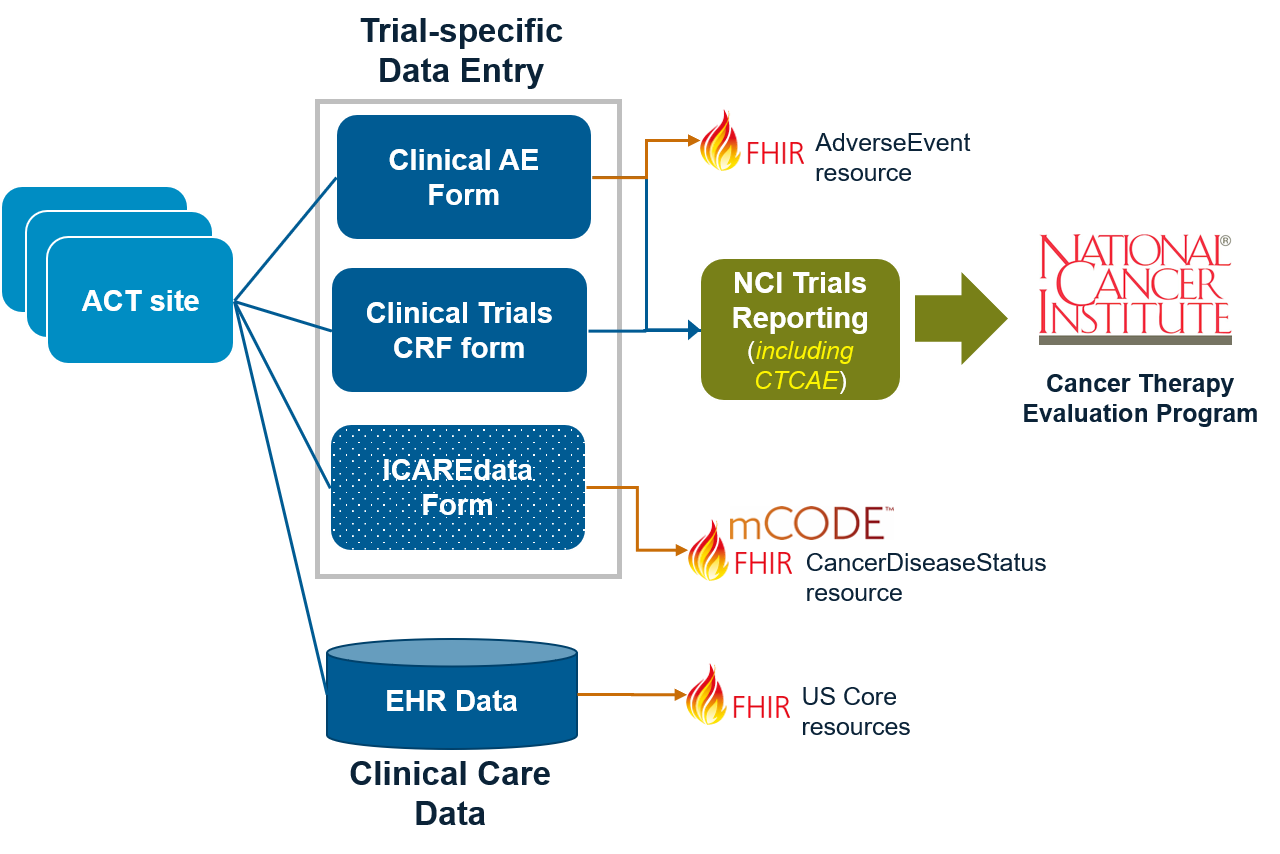
Home - HL7 FHIR Implementation Guide: Common Terminology Criteria (CTC) Adverse Events Release 1 - DRAFT v0.0.1

NCI CTEP Clinical Research (@NCICTEP_ClinRes) / X
Where Do Clinical Trials Come From

PPT - Basic Data Collection Elements in Cancer Clinical Trials

Cancer Therapy Evaluation Program - an overview

PPT - Writing Clinical Research Protocols Part 1: Preparation and

ECOG E2100 - Cancer Therapy Evaluation Program (CTEP)

Presenting the 2022 Leonard M. Rosen Memorial Research Award to NCI's Malcolm Smith — Children's Cancer Cause

Branches of the Cancer Therapy Evaluation Program (CTEP) at the