
Crucial Steps for Singapore Medical Device Registration & HSA Approval
4.8 (747) In stock

4.8 (747) In stock
Discover the crucial steps for successful Singapore medical device registration and HSA approval. Operon Strategist offers expert guidance, classification insights, and comprehensive support. Contact Operon Strategist to learn more and navigate the regulatory landscape with confidence.
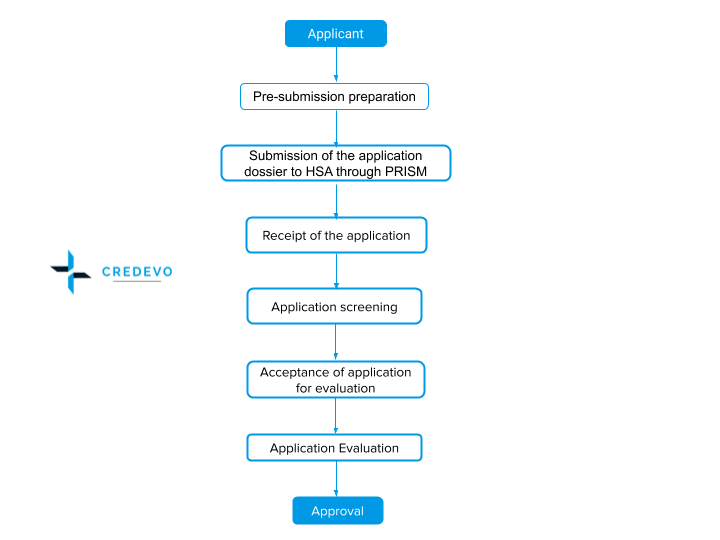
Generic Drug Registration Process In Singapore

Academic Report on Singapore HSA Class D and Australia TGA Class III Medical Device Regulatory Overview & Strategy
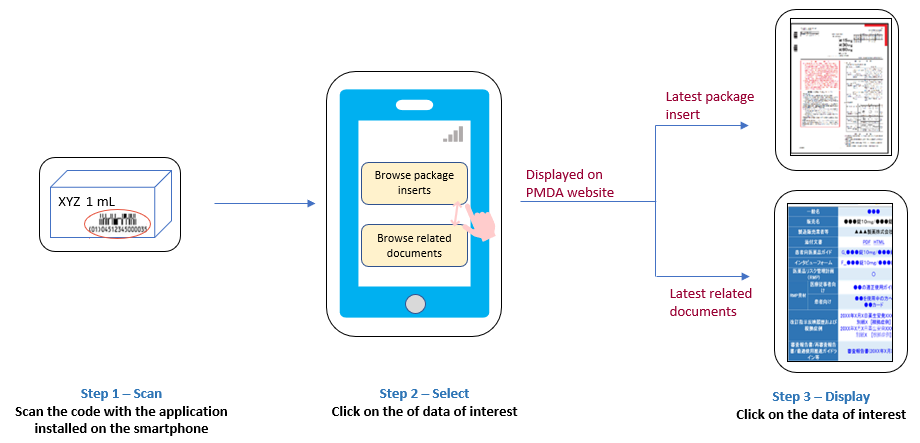
E-labeling and digital transformation in healthcare

Academic Report on Singapore HSA Class D and Australia TGA Class III Medical Device Regulatory Overview & Strategy
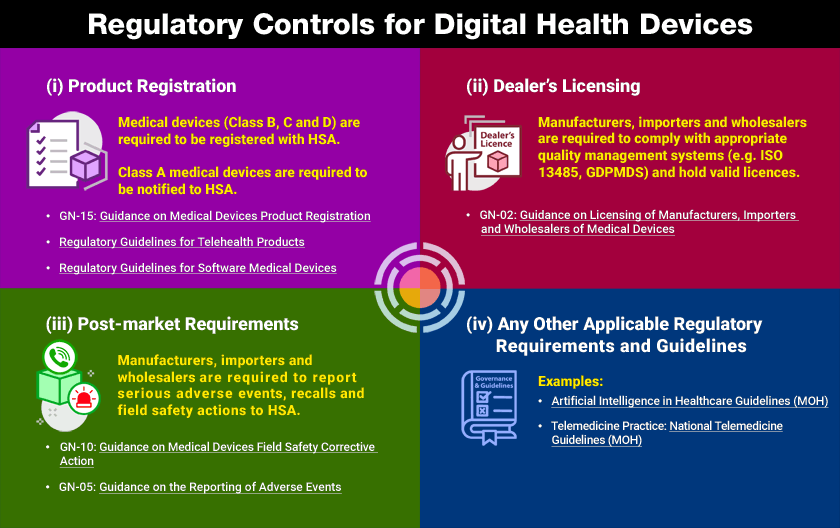
HSA Digital Health

/images/default-source/logo/csa_ihis

Medical Device Regulations Representation for Singapore

Medical Device Registration in United States of America
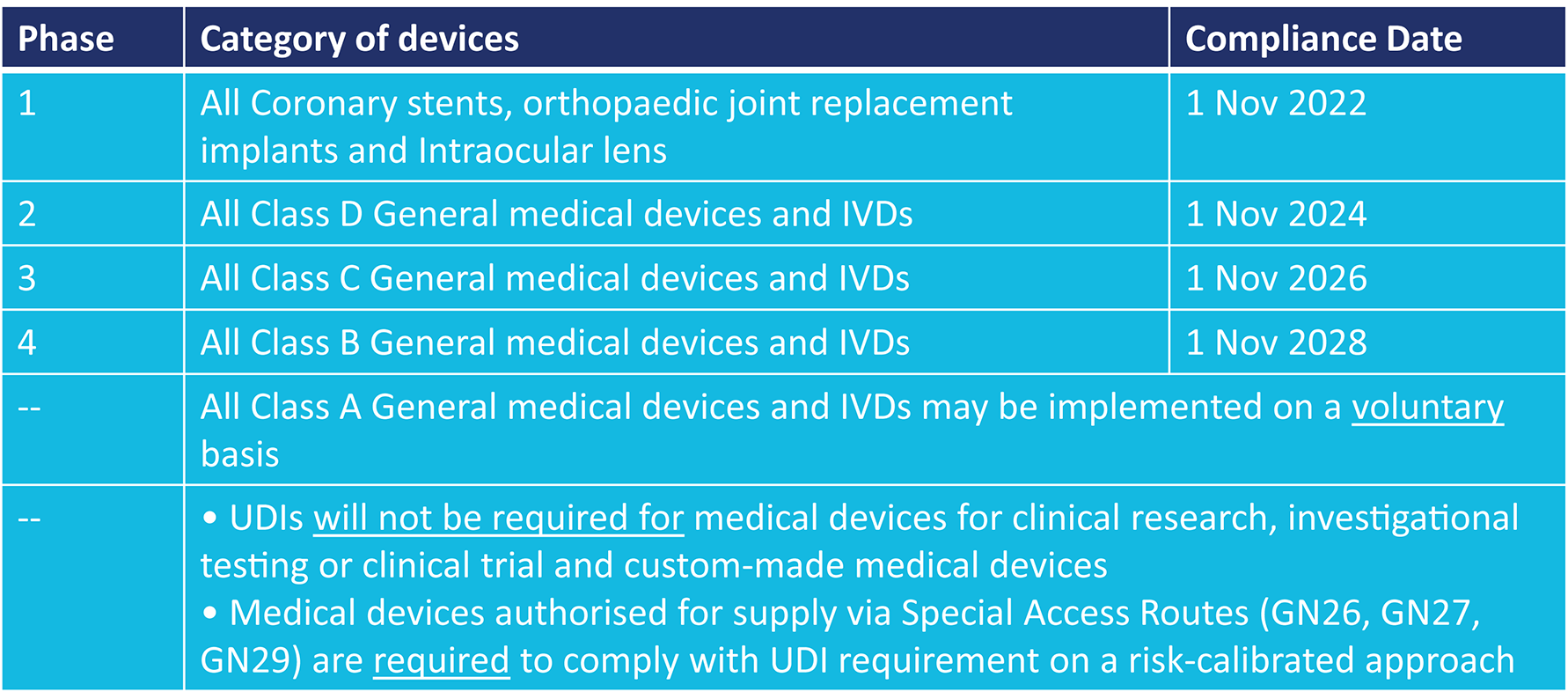
Singapore HSA Published Guidance on UDI System

Singapore's HSA - Global Regulatory Partners, Inc.

理工科技顧問股份有限公司 Qualtech Consulting Corporation

PDF) REGISTRATION AND APPROVAL PROCESS OF MEDICAL DEVICES IN INDIA AND SINGAPORE QR code
UDI overview in Asia - Europe IT Consulting

Medical Device Registration in Singapore